GreyMatters
Educational articles, BetterBrain resources, latest in brain health, and news about us. Written for you, with expertise (and love).

Visceral Fat and the Brain: What 16 Years of Data Actually Show
A study just landed in Nature Communications that reframes how to think about midlife weight loss and brain health. Researchers followed 533 adults for up to 16 years after they completed lifestyle interventions, then scanned their brains and tested their cognition. The finding: it wasn't weight loss that predicted slower brain atrophy and better cognitive scores years later. It was sustained loss of visceral fat specifically.
This matters because most people, and most doctors, still track weight on a scale. The number on the scale lumps together fat, muscle, water, and where on the body the fat actually sits. The new data suggests that for brain health, where the fat is matters more than how much of it there is.
What the study found
The research, called the Follow-Interventions-Trials (FIT) project, pulled participants from four earlier 18 to 24 month lifestyle randomized trials. The average age at follow-up was 61. Each participant had abdominal MRI, brain MRI, and Montreal Cognitive Assessment (MoCA) testing 5 to 16 years after their original intervention.
Three findings stood out. Lower long-term visceral fat exposure, calculated across baseline, post-intervention, and follow-up, independently predicted higher cognitive scores. Visceral fat loss during the intervention period predicted higher brain volumes years later, independent of overall weight loss. And among the participants who had three full sets of scans over the years, lower long-term visceral fat was associated with a slower rate of brain atrophy.
The same patterns were not observed for subcutaneous fat, the kind that sits under the skin and shows up on a pinch test. Visceral fat, the deeper fat surrounding the organs, was the variable that mattered.
The proposed mechanism is glycemic. Visceral fat is metabolically active. It secretes inflammatory signals and contributes to insulin resistance. Both of these are increasingly understood as drivers of cognitive decline. When visceral fat goes down, glycemic control improves, and the brain appears to benefit downstream.
Why this changes how we think about midlife weight
Most people who decide to lose weight in their fifties do not distinguish between subcutaneous fat, visceral fat, and muscle. Many lose all three. Losing muscle in midlife is a problem on its own. Losing subcutaneous fat without losing visceral fat is largely cosmetic. Losing visceral fat appears to be the part that actually protects the brain.
This also reframes "skinny fat." A person can have a body mass index in the normal range and still carry significant visceral fat. The scale will not flag this. A waist measurement, a DEXA scan, or abdominal imaging will.
For anyone in their 40s, 50s, or 60s thinking about brain health, the practical question is no longer "should I lose weight." It is "what is my visceral fat doing, and what is actually moving it."
What you can do
Three things tend to move visceral fat without requiring dramatic restriction.
The first is reducing refined carbohydrates and added sugars. Visceral fat is more responsive to insulin signaling than subcutaneous fat. Lowering postprandial glucose spikes is associated with reduced visceral fat over time.
The second is consistent moderate-intensity movement, especially after meals. A 10 to 15 minute walk after eating blunts the post-meal glucose curve. Repeated over months, this contributes meaningfully to visceral fat reduction.
The third is strength training, which we covered last week. Muscle is the primary site of glucose disposal in the body. More muscle means better glycemic control, which means less visceral fat accumulation.
The biomarkers that respond, and the ones we look at first with members, are HbA1c (your average blood sugar over three months), fasting insulin, and the broader metabolic panel that looks at lipids and liver function. These shift before the scale shifts. They tell you whether the metabolic conditions that drive visceral fat are improving.
The bottom line
The next time someone tells you to lose weight for your brain, the more useful version of that advice is to lose visceral fat for your brain. The number on the scale was always a poor proxy for what is happening inside the body. The new research makes that clearer.
If you are already a BetterBrain member, your most recent HbA1c, fasting insulin, and metabolic panel results are in your dashboard. Worth a fresh look in light of this.
If you have not yet started, these markers are part of the BetterBrain Essentials Panel we look at first.

Strength Training and Brain Health: What 6 Months of Lifting Does to Your Hippocampus
Strength Training and Brain Health: What 6 Months of Lifting Does to Your Hippocampus
Aerobic exercise gets most of the brain-health attention. The cardiovascular benefits are well-documented, the GPLD1 pathway connecting movement to blood-brain barrier repair is real, and almost every brain health protocol recommends it.
But a body of research has been quietly building on a different type of exercise entirely. And a comprehensive 2025 network meta-analysis just made the case for strength training impossible to ignore.
What the research found
The analysis, published in Frontiers in Aging Neuroscience, pulled data from dozens of randomized controlled trials involving cognitively healthy older adults. When researchers compared aerobic exercise, resistance training, mind-body practices like yoga, and combined programs for their effect on cognitive function, resistance training produced the strongest overall effect on global cognition. The effect size was considered moderate to large in cognitive research, where meaningful improvements are notoriously difficult to achieve.
A parallel 2025 systematic review went further. It examined actual brain imaging alongside cognitive testing. The findings: at least two resistance training sessions per week, sustained for at least six months, were associated with measurable increases in cortical thickness in two critical brain regions.
The hippocampus, your brain's primary memory formation center. And the prefrontal cortex, involved in planning, complex reasoning, and self-control. These are precisely the regions most vulnerable to aging and most associated with early cognitive decline.
These weren't just improvements in test scores. These were structural changes in brain tissue, visible on MRI scans. Six months of consistent strength training literally changed the physical structure of participants' brains.
How it works
The mechanisms connecting resistance training to brain health are distinct from aerobic exercise benefits, which is exactly why both matter.
When your skeletal muscles contract under load, they secrete signaling proteins called myokines into your bloodstream. One well-studied myokine, irisin, crosses the blood-brain barrier and has been shown to increase new brain cell formation and reduce neuroinflammation in research models.
Resistance training also improves insulin sensitivity in your muscles, contributing to better metabolic health throughout your body. Given that brain insulin resistance is increasingly understood as a central mechanism in Alzheimer's disease, this metabolic pathway from strength training to brain protection isn't minor.
The exact dose the research supports
Frequency: At least two sessions per week. Some studies suggest three sessions produce greater effects, following a dose-response pattern.
Duration: Six months minimum for structural brain changes to appear on imaging. This isn't a quick fix. It's a sustained practice.
Intensity: Moderate intensity works best, approximately 50 to 70 percent of your maximum. In practical terms, this means lifting a weight you can complete 8 to 12 controlled repetitions with, where the final 2 to 3 reps require genuine effort.
Going through the motions with very light resistance doesn't appear to produce the same stimulus. The dose matters as much as the activity itself.
You don't need a gym membership to start. Bodyweight exercises (squats, push-ups, lunges), resistance bands, or even filled water bottles work. The goal is progressive challenge over time, gradually increasing difficulty as you get stronger.
Tracking whether it's working
Several markers respond to consistent strength training and give you objective evidence the work is paying off.
Insulin sensitivity: Resistance training improves how your cells respond to insulin. You can track this through fasting insulin and HbA1c, which shows your average blood sugar control over three months. Better insulin sensitivity is associated with better cognitive function.
Inflammatory markers: Consistent strength training reduces systemic inflammation. hs-CRP and homocysteine levels respond, giving objective evidence of the anti-inflammatory effect.
Metabolic health markers: Strength training improves multiple metabolic markers including glucose control, lipid profiles like VLDL-C, and overall metabolic function. These improvements happen through muscle-mediated pathways that complement what aerobic exercise does.
Seeing these numbers move is concrete evidence that the effort is producing real change.
The case for combining aerobic and resistance training
Aerobic exercise works through the GPLD1 pathway, where your liver releases protective signals during movement that repair your blood-brain barrier.
Strength training works through entirely different pathways: muscle-derived signaling proteins, improved insulin sensitivity, and hormonal changes that protect brain structure.
These are complementary, not redundant. Research on combined programs shows that doing both produces greater cognitive benefit than either alone. If you're already doing aerobic exercise regularly, adding just two strength sessions per week hits the research-backed minimum.
If you're curious where your insulin sensitivity, inflammation, and metabolic markers stand, BetterBrain's Blueprint testing covers all of them in a single panel, plus 40 more brain health markers.
Cholesterol and Your Brain: What You Actually Need to Know
Cholesterol gets a bad rap. For decades, we've heard that it clogs arteries and causes heart attacks. But here's what doesn't make headlines: your brain contains 25% of your body's total cholesterol despite making up only 2% of your body weight. As lipid expert Dr. Tom Dayspring puts it, "Cholesterol is almost certainly the most important molecule in the brain."
So how can cholesterol be both necessary for brain function and a major cardiovascular risk factor? The answer lies in understanding where cholesterol is, how it gets there, and what form it takes.
In this post, we're taking a deeper dive into cholesterol than usual. We'll break down what your cholesterol numbers actually mean, why the standard tests miss critical information, and how the cholesterol story connects cardiovascular health to brain health.
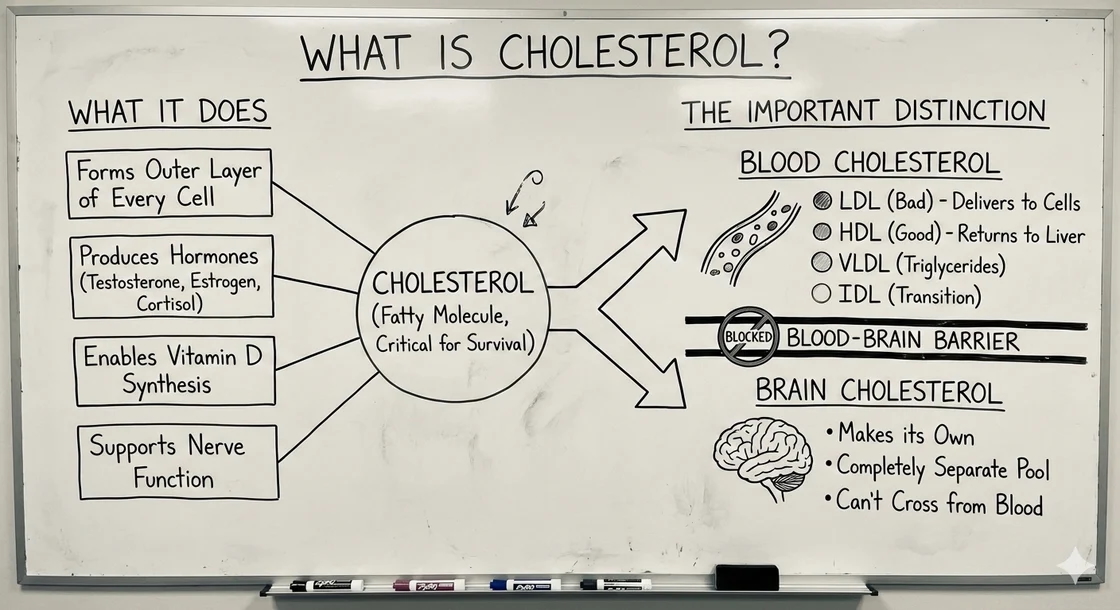
The Basics: What Is Cholesterol?
Cholesterol is a fatty molecule that serves critical functions throughout your body. It forms the outer layer of every cell, helps produce hormones (including testosterone, estrogen, and cortisol), enables vitamin D synthesis, and supports nerve function. Without cholesterol, you simply couldn't survive.
But there's an important distinction to make: "cholesterol" isn't one thing.
When we talk about cholesterol in the blood versus cholesterol in the brain, we're really talking about two separate systems that operate differently.
Blood cholesterol: Cholesterol travels through your bloodstream inside particles called lipoproteins. These particles come in different types:
- LDL (low-density lipoprotein): Often called "bad" cholesterol, LDL particles deliver cholesterol to cells throughout your body
- HDL (high-density lipoprotein): Called "good" cholesterol, HDL particles help transport excess cholesterol back to the liver
- VLDL (very low-density lipoprotein): Primarily transports triglycerides but also carries some cholesterol
- IDL (intermediate-density lipoprotein): A transition form between VLDL and LDL
Brain cholesterol: Your brain makes its own cholesterol and keeps it completely separate from blood cholesterol. The blood-brain barrier prevents cholesterol in your bloodstream from entering your brain. This means the cholesterol circulating in your blood and the cholesterol in your brain are part of two entirely different pools.
The Heart Risk: Why apoB Matters More Than LDL?
What is apoB?
ApoB (apolipoprotein B) is a protein found on the surface of all atherogenic (plaque-forming) particles, including LDL, VLDL, IDL, and Lp(a). Each particle contains exactly one apoB molecule, making apoB a direct count of total potentially harmful particles.
Why apoB beats LDL cholesterol?
Traditional LDL-C (LDL cholesterol) tells you how much cholesterol LDL carries, but not how many particles you have. Two people can have identical LDL-C but vastly different particle numbers and risk levels.
- Many small LDL particles (each carrying less cholesterol) = "normal" LDL-C but high particle count and high risk
- Fewer large LDL particles (each carrying more cholesterol) = higher LDL-C but fewer particles and potentially lower risk
ApoB counts every atherogenic particle regardless of size. This makes it the best single marker for cardiovascular risk.
What is the triglyceride connection?
Elevated triglycerides (often from insulin resistance) fundamentally alter lipid metabolism:
- VLDL becomes triglyceride-enriched
- Particles get remodeled into small, dense LDL
- ApoB particle count increases
- You can have "normal" LDL-C but dangerously high apoB
This is why metabolic health is critical for cardiovascular risk.
The Brain Side: Why Cholesterol Is Essential
While high apoB threatens your heart, brain cholesterol is absolutely essential.
Critical brain functions
- Myelin formation: Insulates nerve fibers for rapid signal transmission
- Synapse formation: Creates and maintains neuron connections
- Cell membranes: Every neuron needs cholesterol-rich membranes
- Neurotransmitter release: Regulates how neurons communicate
How the brain gets cholesterol
Astrocytes (brain cells) produce cholesterol and package it into brain-specific lipoproteins containing apoE (apolipoprotein E) for delivery to neurons.
The APOE genetic factor
- APOE ε2: Protective against Alzheimer's, better cholesterol handling
- APOE ε3: Most common, considered neutral
- APOE ε4: Increases Alzheimer's risk, disrupts brain cholesterol metabolism
Research from MIT shows APOE4 is associated with brain cells accumulating cholesterol abnormally rather than using it to make healthy myelin. This isn't about too much or too little cholesterol, but how effectively the brain uses it.
Cholesterol and Alzheimer's
Brain regions vulnerable to Alzheimer's show signatures of being "super cholesterol-hungry," as researchers describe it, constantly trying to produce and absorb cholesterol. When this system fails (especially with APOE4), neurodegeneration may follow.
The Paradox Resolved
In the bloodstream: High apoB drives atherosclerosis. Particles penetrate artery walls, oxidize, trigger inflammation, form plaques. Goal: Keep apoB low (<60-80 mg/dL) to prevent cardiovascular disease.
In the brain: The brain makes its own cholesterol independently. Blood cholesterol can't cross the blood-brain barrier. Goal: Support healthy brain cholesterol metabolism through metabolic health.
This separation means lowering blood cholesterol doesn't starve your brain. Your brain continues making what it needs regardless of apoB levels.
The Bottom Line
Cholesterol isn't inherently good or bad; context is everything. In your bloodstream, high apoB poses serious cardiovascular and cerebrovascular risks. In your brain, cholesterol is essential for structure and function.
The good news: these systems are separate. Lowering apoB to protect your heart doesn't harm your brain. In fact, protecting your cardiovascular system through better lipid management, metabolic health, and inflammation control also protects your brain.
Understanding which biomarkers matter empowers informed decisions. It's not about fearing cholesterol; it's about managing it intelligently.
Want to Understand Your Complete Lipid Profile and Genetic Risk?
BetterBrain includes apoB, advanced lipid testing, metabolic biomarkers, and APOE genetic testing for a complete cardiovascular and brain health picture.

Unlocking the Power of Omega-3 Fatty Acids
As the quest for effective Alzheimer’s prevention continues, recent studies1,2 underscore the profound impact of dietary choices on our brain health. One nutrient class consistently at the forefront of neuroprotective research is Omega-3 fatty acids, particularly Docosahexaenoic Acid (DHA) and Eicosapentaenoic acid (EPA). In this post, we explore how integrating DHA and EPA into your diet can play a crucial role in reducing the risk of Alzheimer's disease.
Fatty acids explained1
DHA and EPA are a major omega-3 fatty acid predominantly found in fish oils. They are called essential fatty acids, meaning that the human body cannot produce them on its own, so they must come from dietary sources. Make sure not to confuse omega-3s (like DHA and EPA) with omega-6s. These are a different class of fatty acids typically found in vegetable oils and nuts. They are much more common in most diets, and can promote inflammation when consumed in excess.
DHA and EPA are particularly important because the molecules are building blocks for neurons. This means that maintaining healthy levels of DHA and EPA supports neuronal membrane integrity, promotes healthy synaptic activity, and mitigates inflammation within the brain. Long-term, these fatty acids have been shown to help preserve cognitive abilities and delay the onset of dementia1.
Clinical insights on Alzheimer’s prevention
A growing body of research points to a direct correlation between DHA and EPA intake and a reduction in the risk for Alzheimer's. Animal studies provide compelling evidence, showing that diets rich in DHA can significantly reduce the formation of amyloid plaques, which are closely linked to Alzheimer’s pathology1.
Furthermore, in human epidemiological research (research that investigates the distributions and determinants of health-related events in populations), increased consumption of DHA through dietary sources like fish has been associated with lower incidence rates of Alzheimer’s, suggesting its significant protective effect. One study3 found that people with the highest levels of DHA had a 49% lower risk of developing Alzheimer's disease compared to those with the lowest levels. This means that those in the top 20% were about half as likely to get Alzheimer's as those in the bottom 20%. Additionally, increasing DHA levels from the lowest group to the highest group was predicted to give an extra 4.7 years of life free from Alzheimer's disease.
Other studies have concluded similar results, showing a 47%5 reduction in risk, though there is debate about when this effect may occur. The consensus is that DHA supplementation is most effective when started early, before symptoms get classified as dementia6.
Increasing your omega-3 intake
There are two main ways to make sure you’re getting enough DHA and EPA - either making conscious dietary choices4 or taking supplements. From the diet side, fatty fish are an excellent source of omega-3s. Typical guidance recommends eating fish like salmon, mackerel, herring, or halibut at least 3 times per week. It’s worth noting that chia seeds and flax seeds are also excellent sources of omega-3s. Predatory fish like tuna are also good sources of EPA and DHA, but be careful not to consume them too often since they also contain high levels of mercury.
Fish oil supplements can be another great way to increase your intake of omega-3s - if you carry the APOE4 gene, supplementation is particularly important since you may have more trouble absorbing dietary omega-3s. However, it’s important to recognize that not all supplements were created equal. Specifically, not all brands will have the same purity of fatty acids, and not all will contain sufficient levels of DHA and EPA. Here are some things to look out for to make sure you are getting high quality fish oil:
- High amounts of DHA and EPA per serving (at least 500mg combined per serving)
- Minimal additives in the ingredients other than the oil and capsule contents
- Packaged in dark containers to protect the oil from light
A Step Towards Cognitive Longevity
Embracing a diet that includes adequate amounts of DHA and EPA can significantly contribute to brain health and potentially decrease the risk of Alzheimer’s. Start by evaluating your current dietary habits and consider how you might improve your omega-3 intake, ensuring your brain remains vibrant and healthy well into later life.
Where to get started
- Measure your blood omega-3 index to learn your current EPA and DHA levels. This is one of the 50+ biomarkers tested during your BetterBrain Essentials blood draw.
- Eat at least 3 servings of fatty fish (e.g., salmon, mackerel) per week, but make sure not to overdo your intake of predatory fish like tuna.
- Consider using fish oil supplements to increase your EPA and DHA intake. We recommend Carlson’s fish oil, which is available at a discount with a BetterBrain membership.
The Role of Homocysteine in Dementia Risk
Homocysteine is a naturally occurring amino acid in our body and can spike acutely, such as after staying up all night. In healthy people, homocysteine naturally clears over time. However, at chronic high concentrations, it is associated with various health issues, including heart disease and, notably, cognitive decline. In fact, having blood homocysteine levels over 14 μmol/L is associated with a nearly doubled risk of dementia1. Luckily, there are simple ways to lower your homocysteine levels - most notably B vitamin supplementation.
Brain Atrophy, Aging and Cognitive Decline
As we age, our brains naturally undergo some amount of atrophy, or decrease in size, which involves a loss of neurons and their connections. This process is accelerated in Alzheimer’s dementia2. With a lower brain volume and fewer neural connections, it’s easy to see how atrophy can lead to lasting cognitive impairment. One factor that has a strong influence on the rate of brain atrophy is blood concentration of homocysteine. Several studies3,4 link elevated homocysteine levels are linked with a heightened risk of dementia.
The role of B vitamins
You won’t often hear us say this, but you have an ace up your sleeve in fighting homocysteine: B vitamins. One study2 investigated the effects of using B6, B9, and B12 vitamins over the course of two years, using the same level of rigor that is commonly used for drug clinical trials.
On average, people who used B vitamins lowered their overall homocysteine levels by 32% and experienced a 30% slower rate of brain atrophy. In fact, those who started with very high levels of blood homocysteine (> 14 μmol/L) managed to slow their atrophy rate by 53%. In other words, this study suggests that the simple act of taking a daily B vitamin supplement can cut your dementia risk in half.
The type of B vitamin matters
There are two factors to consider when selecting a B vitamin supplement. The first is what vitamins you are getting. The study mentioned above specifically tested the use of vitamins B6, B9 (also known as folate), and B12. It’s important to get a mix of both. The second is whether or not you select the methylated form of the vitamins. Methylation is a biological process that makes the vitamins more available for your body to use. In other words, the same dose of methyl-B vitamins will be more strongly absorbed than normal B vitamins. This is especially important for people with mutations in the MTHFR gene, since they otherwise have trouble absorbing B vitamins. We generally recommend taking methylated B vitamins since they are perfectly safe, but if you are sensitive to overmethylation, you may want to consider regular B vitamins to avoid side effects like headaches, anxiety, or irritability.
Broader Implications for Dementia Prevention
Homocysteine is a critically important risk factor for dementia. Not only does it accelerate brain atrophy, it also aggravates other conditions through inflammation and oxidative stress. Thankfully, B vitamins are an extremely effective tool to lower homocysteine levels. While there are many other ways of managing your homocysteine levels, most notably through diet, exercise, and stress management, B vitamins are a low-effort high-impact way to keep your brain atrophy at bay.
Get started on managing your homocysteine
- Measure your blood homocysteine levels to learn where you stand. Homocysteine is one of the 50+ biomarkers tested during your BetterBrain Essentials blood draw.
- Consider using B vitamin supplements to lower your homocysteine, ideally below 9 μmol/L. We recommend Pure Encapsulations MethylAssist, but make sure you use an unmethylated alternative if you are sensitive to overmethylation.
- Learn more about homocysteine on the Peter Attia Drive episode on dementia. This episode covers many topics, so if you’re just interested in homocystine, skip ahead to 1:09:00.

Rapamycin: Revolutionizing Alzheimer's Prevention?
In the 1960s, researchers on Easter Island were investigating local indigenous peoples’ claims that the soil has healing properties. After testing various soil samples, the researchers isolated a small molecule they believed was responsible for the effects. They named it rapamycin5, after the traditional name for the island, Rapa Nui. Since its discovery, rapamycin has been used in various settings, from an antifungal agent to more recently a beacon of hope in anti-aging medicine. Additionally, it is showing promise in extending lifespan and preventing neurodegenerative diseases such as Alzheimer’s.
From anti-rejection med to longevity enhancer
The transition from an antifungal to a potential longevity drug has been intriguing. Rapamycin, approved in 1999 for its immunosuppressant qualities, is still commonly used in kidney transplants to prevent organ rejection. However, a 2014 study1 on older adults revealed a paradox: at much smaller doses, rapamycin boosted the immune response to flu vaccinations, despite their age-related weakened immune function. This unexpected enhancement suggests that rapamycin might have broader applications for disease prevention in older adults, potentially making it a valuable tool in combating age-related declines in the human immune system.
How rapa works
To understand how rapamycin works, it’s important to understand the molecule that it targets: a cellular receptor named mTOR. mTOR is present in nearly all cells in the human body and is responsible for mediating pathways that regulate cell growth, metabolism, and survival. Inhibiting mTOR completely is catastrophic - it prevents cells from making energy, eventually leading to their death. However, partial inhibition means that mTOR has a harder time forming a cluster with other proteins, which makes the cell act as if it's not getting enough food. This starts a process where the cell breaks down unneeded or damaged parts, like proteins that aren't folded correctly, which can otherwise impair the cell’s ability to function. This leads to improved cellular survival and resilience and is particularly relevant to brain health since one of the main features of Alzheimer’s is the accumulation of misfolded amyloid beta protein plaques in neurons.
The partial inhibition of mTOR therefore shows potential for slowing down the progression of Alzheimer’s disease and improving the survival rate of neurons. Animal studies2,3 suggest that rapamycin may help mitigate or improve many of the pathologies associated with Alzheimer's disease and potentially restore cognitive function.
Growing evidence yet limited human trials
While animal studies have robustly demonstrated lifespan and healthspan extensions—with remarkable outcomes like a 20-30% increase in the lifespan of mice4—human data remains scarce. The gap in human trials can be largely attributed to the fact that rapamycin use for longevity is considered “off-label”. This means that the Federal Drug Administration (FDA) has not yet approved rapamycin for this use, which diminishes incentives for comprehensive research funding. However, the evidence from animal studies across a variety of species strongly suggests a significant potential for rapamycin in anti-aging treatments.
Rapamycin’s mainstream use
As mentioned earlier, the FDA has only approved rapamycin for use as an immunosuppressant. It’s important to recognize that rapamycin use for longevity is still considered experimental and will not be reimbursed by insurance. Because the FDA hasn’t established guidelines around its use, there is no single accepted protocol for rapamycin dosage. Given the drug’s complex effects, the risk of experiencing side effects is real. Nonetheless,many individuals already use rapamycin “off-label” under the supervision of a physician for its longevity benefits. This is a perfectly legitimate use of the molecule, even though there is a lot we have yet to learn.
A promising option for the future
Rapamycin offers a compelling glimpse into the future of longevity and neuroprotection. Although its journey from a soil sample to a potential anti-aging miracle has been gradual, the promise it holds could change the landscape of preventive health. As research continues, both the medical community and potential users must weigh the benefits against the uncertainties of translating animal model successes to human health outcomes.
Learn more
Listen to the Peter Attia Drive episode on rapamycin to learn more about the molecule, its history, how it works, and the most recent evidence on its effects.
Get immediate insights with a 3 minute assessment
Start nowFighting Dementia with CBT: A proactive approach
Cognitive-behavioral therapy (CBT) has long been recognized as a powerful tool in managing mental health disorders, but its potential role in preventing dementia is only beginning to be appreciated1,2. There are two ways that CBT can help. First, CBT is a powerful tool which can combat anxiety, depression, and other mental health disorders, which increase the risk of dementia3,4. Second, CBT can be used to facilitate habit change - that is to say, help users adopt behaviors which promote brain health.
How does CBT work?
CBT is a structured form of psychotherapy that relies on the principle of identifying and changing certain thought patterns and behaviors. The underlying principle is that the brain is “plastic” or flexible throughout life, which means that we can shape the connections our brain makes. The therapy is collaborative, with the client and therapist working together to set goals and practice new skills. The specifics of a CBT session will vary depending on the needs of each individual, but sessions generally involve recognizing distorted thinking, challenging irrational beliefs, and developing healthier responses. CBT also encourages behavioral changes by gradually facing fears or engaging in positive activities.
CBT in Alzheimer’s prevention
Combating mental illness
Your mental health is closely linked to your brain health. Depression, anxiety, and other disorders can actually increase inflammatory stress in your brain and disrupt your cognitive function. There are several different approaches to treating mental illness, and CBT happens to be highly effective for some people.
Forming healthy habits
Much of dementia prevention relies on addressing known lifestyle risk factors, things like physical inactivity, poor diet, diabetes, and high blood pressure. Forming and maintaining healthy habits can feel challenging, but CBT can help facilitate behavior change and keep you on the right track.
Common CBT principles
Because CBT is a tool, many of its principles apply just as well to managing mental health as to improving lifestyle habits. Common techniques include:
- Awareness and Identification: CBT begins with helping individuals become more aware of their current state. What situations or thoughts trigger a negative cascade? Which of my habits aren’t the healthiest?
- Cognitive Restructuring: This is a fancy way of saying challenging negative thoughts. By examining negative emotions and thoughts, individuals can start to think about them objectively. For mental health, this can be changing “something catastrophic will happen” to “I can take on this challenge.” For habit formation, this may be replacing "changing my diet won’t make a difference" with "every healthy meal contributes to my resilience."
- Behavioral Experiments: CBT employs behavioral experiments to challenge negative beliefs and test out new behaviors in a controlled manner. This may look like gradually facing a feared situation, engaging in activities that carry a sense of accomplishment, or starting to exercise with 10-minute daily walks. The purpose here is to gain confidence and reinforce the challenge to negative thoughts with real-life actions.
- Goal Setting: Together with a therapist, individuals set specific, achievable goals related to their needs. These may be thing like developing problem-solving skills to improve their sense of control, or things like increasing weekly physical exertion. The most important thing is for these goals to feel realistic and manageable.
- Reinforcement: Positive changes are reinforced through self-monitoring and resilience techniques. Tracking change over time helps establish a feedback loop where improvement becomes a natural motivator, while practical skills for managing setbacks help sustain long-term behavior change.
By systematically working through these steps, CBT provides individuals with the tools and confidence to implement and sustain changes - many of which contribute to preventing dementia.
A strong tool for prevention
It’s important to recognize that CBT isn’t a silver bullet - it’s a tool that’s helped millions of people, and it requires conscious work to implement. With that said, the potential of CBT in preventing Alzheimer’s is vast. It not only helps address mental illness, but also incorporates lifestyle changes that tackle the physiological underpinnings of dementia. Implementing CBT strategies that promote healthy aging could be key in reducing the burden of dementia, emphasizing prevention over treatment and maintaining cognitive vitality well into the later years of life.
Resources on finding CBT providers
If you are interested in trying CBT, there’s several ways to get started.
- Online directories and telehealth platforms can help you find a professional to work with. You can often find therapists through your insurance plan or your employee benefits, or through listing websites like Zocdoc or Psychology Today. You can also look on telehealth platforms like Talkspace or Betterhelp, though not all of these accept insurance.
- Community health centers often also provide CBT services. These are typically available at reduced rates, and focus primarily on mental health.
- Books can help you learn more about CBT and its practice. Feeling Great: The revolutionary new treatment for depression and anxiety is a widely praised book by Dr. David Burns, a psychiatrist and professor of Psychiatry at Stanford.
Harnessing Heart Rate Variability for Brain Health
The intricate dance between our heart rate and brain function is more influential than we might think. Recent studies reveal that heart rate variability (HRV)—the measure of variations between individual heartbeats—affects our brain health, decision-making abilities, and even our emotional regulation. HRV is a physiological metric that we can train ourselves to improve, and its connections to the brain make HRV training a promising technique for dementia prevention and enhancing cognitive function.
Understanding the Heart-Brain Connection:
While your heart rate is the average number of beats per minute, HRV is the difference in length between individual heartbeats.

HRV is not just a measure of heart health - it reflects the health and balance of the autonomic nervous system (ANS), which is responsible for involuntary body functions. In particular, HRV serves as a key indicator of the interaction between the two components of the ANS: the sympathetic (fight or flight) and parasympathetic (rest and digest) nervous systems. Interestingly, there is a two-way feedback system between these nervous systems and the heart. The signals from the sympathetic and parasympathetic influence heart rate, and heart rate regulates the balance of these two systems. By exerting an influence on the ANS, heart rate variability helps regulate emotions and stress responses. In general, higher HRV is associated with better control over decision-making processes, emotional responses, anxiety, and social behaviors1.
Booting your HRV
Common HRV training protocols call for biofeedback, which is a fancy way of saying you need a sensor to measure your heart rate variability so you can improve it. HRV training involves guided exercises such as paced breathing while looking at real-time heart rate data to learn to consciously control heart rate variability. This is what makes it different from techniques like yoga or meditation - their focus is commonly to achieve a balanced state, whereas the goal of HRV training is to modulate physiological responses.
This technique not only promotes a balanced state but also has long-lasting effects on both mental and physical health. Thanks to the heart-brain connection, HRV training leads to improved emotional regulation and stress management. Although different training regimens exist, just five minutes of HRV training twice a day can significantly enhance control over the heart and reduce anxiety levels during stressful periods2. Critically, the benefits of HRV training extend far beyond mood and feeling: they also correlate with enhanced executive functions like planning, problem-solving, and resisting unhealthy impulses1. In fact, the benefits of this training can persist, with improvements seen even 12 weeks after stopping the training1.
Tying back to dementia
Thanks to its influence on stress response, cognitive function, healthy behaviors, and cardiac health, it’s no surprise that higher HRV is associated with stronger resilience against cognitive decline2,3. Since the simple daily practice of HRV biofeedback training can improve your HRV, we can think of it as a valuable tool to help delay the onset and progression of dementia. It's a clear testament to the power of our bodies' interconnected systems, and a reminder that taking care of our heart is as much about our mind.
Where to start with HRV training
- Choose a tracking device: the first step is to make sure you have the right equipment to measure your heart rate variability. Because you are measuring the time between individual heartbeats, you need a particularly sensitive device. Although some common wearables like the Apple watch or Whoop claim to measure HRV, it’s better to opt for a specialized chest strap like the Polar H10 which can connect to your other wearables.
- Visualize your data: once you have a solid device, use an app like Elite HRV or Welltory (both of which have a free version available) so that you can visualize your HRV in real time. These apps also come with a wealth of additional insights that can help you fine tune your training.
- Start training: you can find guided training sessions either through apps like Elite HRV or through online courses provided by organizations like the HeartMath Institute. YouTube also has a number of guided HRV routines you can try, such as this one from the Huberman Lab show.
- Learn more: The Peter Attia Drive podcast has an episode that dives deep into HRV so you can learn more about how it works and how it can help you.
Metabolic Syndrome as a Gateway to Dementia
It’s a well-known fact that Alzheimer’s disease is closely linked to metabolic health, to the point that some experts have begun to refer to it as Type III Diabetes. But what many people don’t know is that poor metabolic health is a strong risk factor for dementia long before it progresses to diabetes, even for people without a family history.
Decoding metabolic syndrome
Before you get diagnosed with diabetes, your body goes through a number of changes. Cells in your body become desensitized to insulin, which usually tells them to absorb sugar from your bloodstream. This results in chronically elevated blood sugar and in turn starts causing problems for your heart, liver, brain, and other organs. This pre-diabetic phase is called metabolic syndrome, and it affects ~25% of adults worldwide, including 40% adults aged 60+ in the U.S. Most of them do not know that metabolic syndrome is a health risk.
Metabolic syndrome is diagnosed when an individual has three or more of the following:
- High triglycerides (≥150 mg/dL)
- Elevated blood pressure (≥130 mmHg systolic or ≥85 mmHg diastolic) OR use of antihypertensive medication
- High fasting glucose (≥100 mg/dL OR use of glucose lowering medications)
- Low HDL cholesterol (<40 mg/dL in men; <50 mg/dL in women OR use of lipid-modifying medication)
- Abdominal obesity (waist circumference ≥102 cm for men and ≥88 cm for women)
It’s worth noting that hemoglobin A1c (HbA1c), a common marker for diabetes, does not feature on this list. Although a doctor won’t use A1c to diagnose metabolic syndrome, it’s still an important marker to keep track of your metabolic health.
Connecting metabolic syndrome to brain health
A large retrospective study1 analyzed data from 175,000 participants over a period of 15 years. Participants were aged 60+ and represented a variety of sociodemographic, lifestyle, and genetic backgrounds. With regards to the metabolic syndrome diagnosis criteria listed above, the study found that:
- Having three conditions increases dementia risk 12%
- Having four to five conditions is associated with an even stronger risk
- The link between metabolic syndrome and dementia is particularly strong in individuals who do not carry the APOE ε4 allele (typically, these are individuals with a lower risk of dementia)
Taking action
The first step is to know where you stand. It’s important to establish a baseline by measuring metabolic markers in your blood and potentially using a continuous glucose monitor (CGM) to see how your blood sugar reacts to your daily habits. This will help you make an informed decision on how to approach your metabolic health.
Metabolic health is strongly tied to your lifestyle - particularly your diet, physical activity, and sleep. There’s also several different drugs, like statins or ACE inhibitors, available to help manage symptoms. However, this post isn’t meant to serve as a meal plan or exercise sheet. Instead, we’ve put together a list of hacks that we think can make a difference even if you haven’t fully optimized your health.
- Drink a tablespoon of vinegar ~20 minutes before a meal. This helps avoid spikes in your blood sugar.
- Go for a walk after meals, especially if those meals are heavy on carbs (e.g., pasta, potatoes, bread). This helps your muscles absorb glucose more efficiently.
- Perform at least 10m of vigorous exercise per day. Raising your heart rate (e.g., through a short HIIT session) helps boost your insulin sensitivity, which keeps blood sugar low.some text
- Exercise is most effective at reducing blood sugar spikes when done within an hour before or after a meal
- Eat your carbs last. Start your meal by eating vegetables (which are high in fiber), then eating proteins and fats, and finally moving to carbs. This helps slow the absorption of sugar into your bloodstream, and in turn reduce sugar spikes.
Small steps add up
Although the hacks mentioned above aren’t meant to replace a healthy lifestyle, they can help you make an immediate improvement in your metabolic health and kickstart your dementia prevention efforts. Remember - working on your metabolic health isn’t just about preventing diabetes, it’s about safeguarding your cognitive health.
Actions to consider
- Measure your metabolic markers. Some of these you may get on your annual physical (e.g., HbA1c, HDL-C, LDL-C, glucose, triglycerides, insulin). Others are less common (e.g., Lp(a), ApoB). Most importantly, don’t settle for just ok. These markers are worth optimizing. All the tests mentioned above are part of the 50+ biomarkers tested during your BetterBrain Essentials blood draw.
- Consider using a CGM to get a more accurate view of how your body processes sugar. Using a CGM, you can observe the effects of what you eat and your lifestyle on your blood sugar in real time, which can help inform the daily choices you make. We love the Dexcom G7, which may be challenging to buy over-the-counter but can be bought for cheaper through Signos.
- Learn more about homocysteine on the Peter Attia Drive podcast #252. This episode covers a wide variety of topics related to brain health, and links many different risks and possible interventions to metabolic health.
Rapamycin: Revolutionizing Alzheimer's Prevention?
In the 1960s, researchers on Easter Island were investigating local indigenous peoples’ claims that the soil has healing properties. After testing various soil samples, the researchers isolated a small molecule they believed was responsible for the effects. They named it rapamycin5, after the traditional name for the island, Rapa Nui. Since its discovery, rapamycin has been used in various settings, from an antifungal agent to more recently a beacon of hope in anti-aging medicine. Additionally, it is showing promise in extending lifespan and preventing neurodegenerative diseases such as Alzheimer’s.
From anti-rejection med to longevity enhancer
The transition from an antifungal to a potential longevity drug has been intriguing. Rapamycin, approved in 1999 for its immunosuppressant qualities, is still commonly used in kidney transplants to prevent organ rejection. However, a 2014 study1 on older adults revealed a paradox: at much smaller doses, rapamycin boosted the immune response to flu vaccinations, despite their age-related weakened immune function. This unexpected enhancement suggests that rapamycin might have broader applications for disease prevention in older adults, potentially making it a valuable tool in combating age-related declines in the human immune system.
How rapa works
To understand how rapamycin works, it’s important to understand the molecule that it targets: a cellular receptor named mTOR. mTOR is present in nearly all cells in the human body and is responsible for mediating pathways that regulate cell growth, metabolism, and survival. Inhibiting mTOR completely is catastrophic - it prevents cells from making energy, eventually leading to their death. However, partial inhibition means that mTOR has a harder time forming a cluster with other proteins, which makes the cell act as if it's not getting enough food. This starts a process where the cell breaks down unneeded or damaged parts, like proteins that aren't folded correctly, which can otherwise impair the cell’s ability to function. This leads to improved cellular survival and resilience and is particularly relevant to brain health since one of the main features of Alzheimer’s is the accumulation of misfolded amyloid beta protein plaques in neurons.
The partial inhibition of mTOR therefore shows potential for slowing down the progression of Alzheimer’s disease and improving the survival rate of neurons. Animal studies2,3 suggest that rapamycin may help mitigate or improve many of the pathologies associated with Alzheimer's disease and potentially restore cognitive function.
Growing evidence yet limited human trials
While animal studies have robustly demonstrated lifespan and healthspan extensions—with remarkable outcomes like a 20-30% increase in the lifespan of mice4—human data remains scarce. The gap in human trials can be largely attributed to the fact that rapamycin use for longevity is considered “off-label”. This means that the Federal Drug Administration (FDA) has not yet approved rapamycin for this use, which diminishes incentives for comprehensive research funding. However, the evidence from animal studies across a variety of species strongly suggests a significant potential for rapamycin in anti-aging treatments.
Rapamycin’s mainstream use
As mentioned earlier, the FDA has only approved rapamycin for use as an immunosuppressant. It’s important to recognize that rapamycin use for longevity is still considered experimental and will not be reimbursed by insurance. Because the FDA hasn’t established guidelines around its use, there is no single accepted protocol for rapamycin dosage. Given the drug’s complex effects, the risk of experiencing side effects is real. Nonetheless,many individuals already use rapamycin “off-label” under the supervision of a physician for its longevity benefits. This is a perfectly legitimate use of the molecule, even though there is a lot we have yet to learn.
A promising option for the future
Rapamycin offers a compelling glimpse into the future of longevity and neuroprotection. Although its journey from a soil sample to a potential anti-aging miracle has been gradual, the promise it holds could change the landscape of preventive health. As research continues, both the medical community and potential users must weigh the benefits against the uncertainties of translating animal model successes to human health outcomes.
Learn more
Listen to the Peter Attia Drive episode on rapamycin to learn more about the molecule, its history, how it works, and the most recent evidence on its effects.
Unlocking the Power of Omega-3 Fatty Acids
As the quest for effective Alzheimer’s prevention continues, recent studies1,2 underscore the profound impact of dietary choices on our brain health. One nutrient class consistently at the forefront of neuroprotective research is Omega-3 fatty acids, particularly Docosahexaenoic Acid (DHA) and Eicosapentaenoic acid (EPA). In this post, we explore how integrating DHA and EPA into your diet can play a crucial role in reducing the risk of Alzheimer's disease.
Fatty acids explained1
DHA and EPA are a major omega-3 fatty acid predominantly found in fish oils. They are called essential fatty acids, meaning that the human body cannot produce them on its own, so they must come from dietary sources. Make sure not to confuse omega-3s (like DHA and EPA) with omega-6s. These are a different class of fatty acids typically found in vegetable oils and nuts. They are much more common in most diets, and can promote inflammation when consumed in excess.
DHA and EPA are particularly important because the molecules are building blocks for neurons. This means that maintaining healthy levels of DHA and EPA supports neuronal membrane integrity, promotes healthy synaptic activity, and mitigates inflammation within the brain. Long-term, these fatty acids have been shown to help preserve cognitive abilities and delay the onset of dementia1.
Clinical insights on Alzheimer’s prevention
A growing body of research points to a direct correlation between DHA and EPA intake and a reduction in the risk for Alzheimer's. Animal studies provide compelling evidence, showing that diets rich in DHA can significantly reduce the formation of amyloid plaques, which are closely linked to Alzheimer’s pathology1.
Furthermore, in human epidemiological research (research that investigates the distributions and determinants of health-related events in populations), increased consumption of DHA through dietary sources like fish has been associated with lower incidence rates of Alzheimer’s, suggesting its significant protective effect. One study3 found that people with the highest levels of DHA had a 49% lower risk of developing Alzheimer's disease compared to those with the lowest levels. This means that those in the top 20% were about half as likely to get Alzheimer's as those in the bottom 20%. Additionally, increasing DHA levels from the lowest group to the highest group was predicted to give an extra 4.7 years of life free from Alzheimer's disease.
Other studies have concluded similar results, showing a 47%5 reduction in risk, though there is debate about when this effect may occur. The consensus is that DHA supplementation is most effective when started early, before symptoms get classified as dementia6.
Increasing your omega-3 intake
There are two main ways to make sure you’re getting enough DHA and EPA - either making conscious dietary choices4 or taking supplements. From the diet side, fatty fish are an excellent source of omega-3s. Typical guidance recommends eating fish like salmon, mackerel, herring, or halibut at least 3 times per week. It’s worth noting that chia seeds and flax seeds are also excellent sources of omega-3s. Predatory fish like tuna are also good sources of EPA and DHA, but be careful not to consume them too often since they also contain high levels of mercury.
Fish oil supplements can be another great way to increase your intake of omega-3s - if you carry the APOE4 gene, supplementation is particularly important since you may have more trouble absorbing dietary omega-3s. However, it’s important to recognize that not all supplements were created equal. Specifically, not all brands will have the same purity of fatty acids, and not all will contain sufficient levels of DHA and EPA. Here are some things to look out for to make sure you are getting high quality fish oil:
- High amounts of DHA and EPA per serving (at least 500mg combined per serving)
- Minimal additives in the ingredients other than the oil and capsule contents
- Packaged in dark containers to protect the oil from light
A Step Towards Cognitive Longevity
Embracing a diet that includes adequate amounts of DHA and EPA can significantly contribute to brain health and potentially decrease the risk of Alzheimer’s. Start by evaluating your current dietary habits and consider how you might improve your omega-3 intake, ensuring your brain remains vibrant and healthy well into later life.
Where to get started
- Measure your blood omega-3 index to learn your current EPA and DHA levels. This is one of the 50+ biomarkers tested during your BetterBrain Essentials blood draw.
- Eat at least 3 servings of fatty fish (e.g., salmon, mackerel) per week, but make sure not to overdo your intake of predatory fish like tuna.
- Consider using fish oil supplements to increase your EPA and DHA intake. We recommend Carlson’s fish oil, which is available at a discount with a BetterBrain membership.
The Rise of Amyloid Blood Tests
The Landscape of Alzheimer's Biomarkers
When it comes to assessing the risk of cardiovascular disease, we have straightforward biomarkers such as ApoB (LDL) and blood pressure which can predict risk fairly accurately. However, the realm of neurodegenerative diseases like Alzheimer's is a lot more messy. For years, researchers and clinicians have depended PET scans or cerebral spinal fluid (CSF) for analyzing biomarkers like amyloid and tau proteins. But these procedures are expensive ($5-10K in the case of PET) or invasive (CSF requires a lumbar puncture), making them impractical for widespread use.
Enter blood amyloid tests, a relatively new but promising diagnostic aimed at solving this problem.
What Are Blood Amyloid Tests and How Do They Work?
As the name suggests, these tests measure the concentration of various amyloid beta proteins in the blood (and sometimes tau as well) as a proxy for amyloid deposition in the brain. While the scientific community continues to debate whether amyloid pathology is a root cause of Alzheimer's disease or merely a symptom of other underlying brain pathologies, there is a clear association between the extent of amyloid presence in the brain and the manifestation of the disease.
One of the original commercially available tests was PrecivityAD by C2N. This test needs to be ordered by a physician and was designed to predict the likelihood of Alzheimer's Disease (AD) pathology by analyzing a few key factors:
- The ratio of two variants of amyloid beta proteins, specifically amyloid beta 42 and amyloid beta 40
- The ApoE variant of the patient
- The age of the individual
By amalgamating these variables, the test attempts to gauge the probability of a positive PET scan. While it's too early to draw any definitive conclusions, there are two primary reasons why looking at this data could be valuable:
- Comprehensive Risk Assessment: The test results can be used in combination with other factors like family history, genetics, metabolic health, vascular health and cognitive testing to assess the overall risk.
- Monitoring Interventions: The score could be used as a dynamic indicator to monitor the effectiveness of steps taken to mitigate Alzheimer's risk. If the score decreases, it suggests that the ratio of amyloid beta 42 to amyloid beta 40 has changed favorably and thus risk has been reduced.
Since then, C2N launched a second generation version of the tests, PrecivityAD21, which accounts for serum tau concentration for additional accuracy. In August of 2023, Quest launched a direct-to-consumer version of the test that does not require physician ordering. While the test is not quite as accurate as C2N in predicting a positive PET, it comes at a materially lower cost - $400 as of the date of this writing.
Applicability: Who Should Consider blood amyloid tests?
According to medical experts, the test should be reserved for those at high risk of developing Alzheimer's, as its sensitivity and specificity are still not entirely understood. The key term here is "pre-test probability." The higher the pre-test probability, the more reliable the test becomes in predicting a positive or negative outcome.
Final Thoughts
The emergence of amyloid (and tau) blood tests is an exciting development in the field of Alzheimer's research and prevention. Although not a complete solution, they do provide an additional layer of information that can potentially enhance our ability to serve people at risk of developing disease. As our grasp of amyloid biomarkers improves, these tests could become integral parts of a broader, more nuanced approach to diagnosis and risk assessment.
Update April 19, 2024
Roche, in collaboration with Eli Lilly, has launched the Elecsys pTau-2172 blood test, which has recently received breakthrough device designation from the FDA. This means it was recognized as an effective tool for diagnosis, and is a critical milestone in how we identify Alzheimer's Disease. This test targets the tau protein pTau-217 and has the unique ability to distinguish AD from other neurodegenerative diseases. This is particularly important given that clinical AD diagnoses often lack sensitivity and specificity. It’s all the more since over half of patients with cognitive impairment remain undiagnosed or incorrectly diagnosed. This advancement promises to enhance early diagnostic accuracy and significantly improve intervention strategies for AD, particularly as global dementia figures are projected to rise sharply by 2050. pTau-217 is now available as an add-on service with BetterBrain.

.png)